QC Design
POC Glucose Test
Is the QC planning process appropriate for POC devices? You bet. See this example application that shows how to plan QC for a glucose device.
Note: at this time this was written, QC Validator was the QC Design software available. Validator has been replaced by EZ Rules 3, which has all the features and capabilities of the earlier version, as well as new enhancements.
A medical technologist who is a laboratory consultant recently attended my course on the Selection, Evaluation, and Control of Analytical Methods which is intended for senior CLS students at UW-Madison. As a teacher, I enjoy having someone with several years experience sit in on the course to stimulate discussion and bring a real world perspective to the class. In discussing her work, which includes consulting with labs on quality management, she brought up a paper by Wandrup [1] that discussed the clinical and analytical requirements and needs of glucose measurements on whole blood. She wanted to see how the information in this paper could be used with our QC planning process to select QC procedures that would be appropriate for point-of-care (POC) applications.
It's always interesting to take a paper from the literature and show how this QC planning process should work using someone else's numbers, so I used this information with an earlier paper by Woo et. al [2] that presented method performance data on a point-of-care analyzer. This made an especially good example for the students because it brought together the recommendations for quality from one paper and the estimates of method performance from another to provide a more complete picture of how to evaluate method performance and manage quality in a POC situation.
- 1. Define the Quality Requirement
- 2. Evaluate the analytical and preanalytical factors
- 3. Enter parameters in computer program
- 4. Obtain clinical OPSpecs chart
- 5. Assess probabilities of rejection
- 6. Select the control rules and numbers of control measurements, N
- 7. Adopt a Total QC Strategy
- 8. Reassess for changes in performance
- For further reading
We'll again follow the steps of the QC planning process.
1. Define the quality requirement.
In a section on analytical goal setting, Wandrup [1] provides a statement of the clinical quality requirement in the following way: "Clinicians sometimes indicate that measured differences of 18-36 mg/dL (1-2 mmol/L) at a level of 90 mg/dL (5 mmol/L) are therapeutically insignificant." This corresponds to what we call a decision interval (Dint) of
(18/90)*100 = 20%
to (36/90)*100 = 40%.
2. Evaluate analytical and preanalytical factors.
The imprecision of the glucose method at the decision level of interest was found in Table 1 of the paper by Woo et. al [2] where the CV was given as 2.44% at a control mean of 5.4 mmol/L. This performance is based on data obtained when personnel in the emergency department operated the device, so it should represent performance under real operating conditions.
The inaccuracy of the method was determined from the regression statistics presented by Woo et. al [2] in Table 3 (not shown here). Based on 527 specimen comparisons between the POC analyzer and a Beckman CX3 Analyzer, the regression line was given as y=-2.19 mg/dL + 1.051x. At a decision level of 90 mg/dL, the systematic error can be calculated to be 2.4 mg/dL or 2.7%.
Within-subject biological variation is given by Wandrup as an SD of 4.7 mg/dL, which would be
(4.7/90)*100 = 5.2% of the decision level of interest .
4. Select a QC ProcedureAs discussed by Woo et al [2], this portable clinical analyzer would be subject to CLIA regulations, therefore 2 control materials need to be analyzed to comply with the CLIA QC guidelines. With the QC Validator 2.0's automatic QC selection, a QC procedure can be selected by clicking the 2 Materials button. |
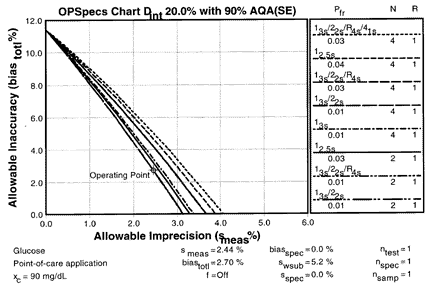 |
5. Review the selected QC procedure.
As shown by the OPSpecs chart generated by the program, a 12.5s rule with N=2 will provide 90% detection of medically important systematic errors, while having a 3% false rejection rate.
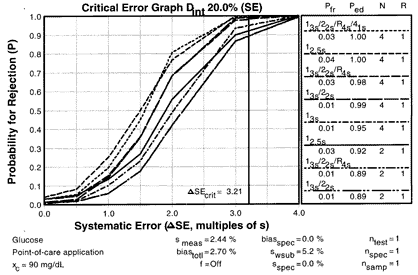
6. Document the performance of the selected QC procedure.In addition to the OPSpecs chart, it is useful to document performance with critical-error graphs for both systematic error and random error. The SE critical-error graph shows that a 13s/22s/R4s multirule procedure will provide nearly as good error detection as the 12.5s procedure and will have a little lower false rejection rate. With the QC Validator program, you can attach these charts to a summary page that documents all the input parameters, calculated parameters, and your final QC selection. |
 |
7. Adopt a total QC strategyThe Total QC strategy should include statistical QC, as well as other QC components such as preventive maintenance, system function checks (e.g., the electronic checks recommended by the manufacturer), and periodic comparisons of performance with other glucose measurement systems. Because 90% AQA can be achieved here with an N of only 2, you can rely on statistical QC and minimize other QC components. It may still be worthwhile to improve method performance; e.g. if method bias were reduced to 1%, then a 13s rule could be used and a false rejection rate of 1% or less achieved with a single rule QC procedure. |
|
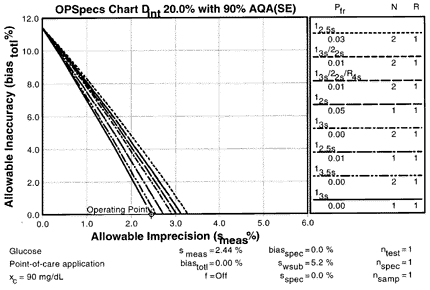
8. Reassess for changesIt is interesting to assess how QC would be affected if the bias of the method were eliminated. For example, if bias were reduced from 2.7% to 0.0%, the OPSpecs chart here shows that the method could be adequately controlled with 1 control measurement per run using 3s control limits, i.e., 13s with N=1. That's would be a simpler and less-costly control procedure. |
 |
Conclusion: Consider a multistage QC strategy
If the bias can be eliminated (through better standardization or calibration), then it would be cost-effective to implement a two-stage QC strategy in which (a) 2 control materials could be analyzed and interpreted with a 12.5s rule whenever there are process changes, such as a new lot of reagents, a new box of reagents brought into service after storage elsewhere, or new less-experience operators, whereas (b) 1 control material could be analyzed and interpreted with a 13s rule to monitor performance during periods of stable operation.
It should be noted that the CLIA proficiency testing criterion for acceptable performance is more demanding and would require use of either 12.5s or 13s/22s/R4s/41s with N=4 to assure that the allowable total error of 10% is not exceeded. Thus, it would actually be desirable to check the test more carefully when a new lot of reagents or major system change occurs. The strategy to use a multistage QC procedure is still applicable, but it may be better to use an N=4 QC procedure to test for the effects of major system changes.
References
- Wandrup JH. Clinical and analytical requirements and needs of glucose measurements on whole blood. Blood Gas News, 1996;5(number 3):3-8.
- Woo J, McCabe JB, Chauncey D, Schug T, Henry JB. The evaluation of a portable clinical analyzer in the emergency department. Am J Clin Pathol 1993;100:599-605.