QC Design
QC for Sysmex Hematology parameters
Based on the Sigma-analysis of the Sysmex xt 1800i, we apply QC Design and analyze the different QC procedures appropriate for different quality requirements.
March 2009
Sten Westgard, MS
This application takes a further look at thea poster presented at last year's IFCC meeting in Fortaleza, Brazil, "Implementation of the process Six Sigma in Haematology's Laboratory" by Recondo C. Grassi C, Blanco M, and Domecq P., Clin Chem Lab Med 2008; 46, Special Suppl, pp S1 – S859, August 2008.
In a previous QC application, Sigma-metrics were calculated for the hematology analytes. In this application, we're going to take those Sigma performance metrics and convert them into recommendations for QC implementation. That is, we're going to design the QC procedures for this instrument based upon the performance of the methods and the quality requirements.
As in the previous study, we're going to present two scenarios: (1) QC Design recommendations based on the use of Biologic-based quality requirements; and (2) QC Design recommendations based on the use of CLIA quality requirements.
Because of the quantitative nature of these decisions, we can no longer use manual tools. So we'll use EZ Rules software to get specific automatic QC recommendations. We'll also use the software to analyze the design differences in error detection and false rejection.
RBC
RBC is an easy analyte to examine. When measured using Biologic-based quality requirements, the Sigma was 4.38. With the CLIA quality requirement, Sigma is 6.33. Both estimates are very good.
Here's the first analysis of RBC, using the Biologic-based quality requirement. The automatic QC recommendation is a 12.5s rule with three controls.
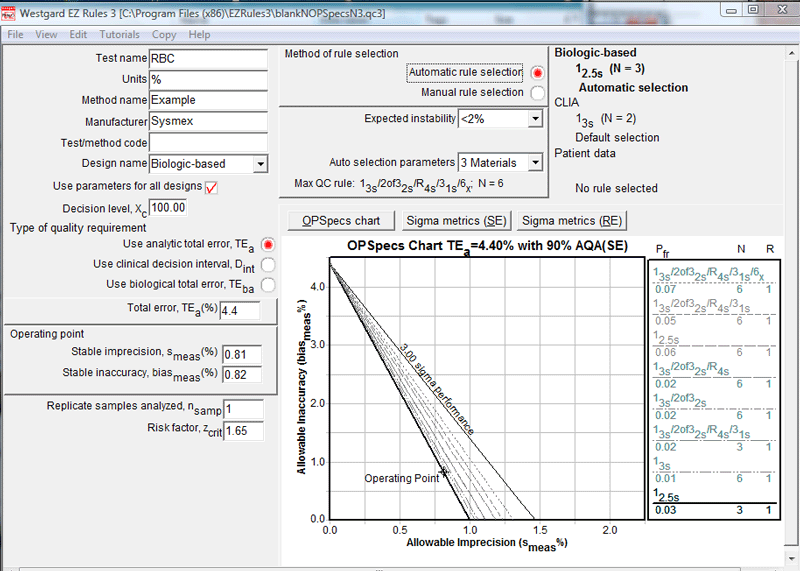
EZ Rules 3 allows you to analyze up to three different quality requirements per analyte. Thus, up on the right corner of the screen, you can see the listing of the three different types. The second analysis, using the CLIA quality requirement, is shown below:
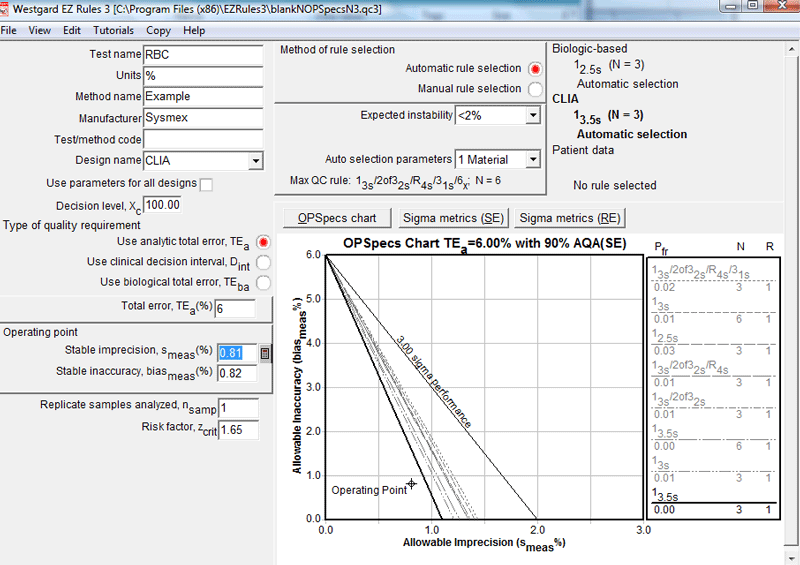
With CLIA, the recommendation is to use 3.5s control limits. This reduces the expected false rejection rate to basically zero.
When the Sigma metrics are on the high end of the scale, the news is good and even better, and the differences are not that significant. Either QC procedure would be acceptable to the laboratory.
HCT
HCT is a more difficult situation. Using the Biologic-based quality requirement, the Sigma is 2.87, while according to the CLIA quality requirement, the Sigma is 4.64. That's a big difference which manifests itself in radically different QC recommendations.
Using the Biologic-based quality requirement, EZ Rules 3 analysis generates this recommendation below:
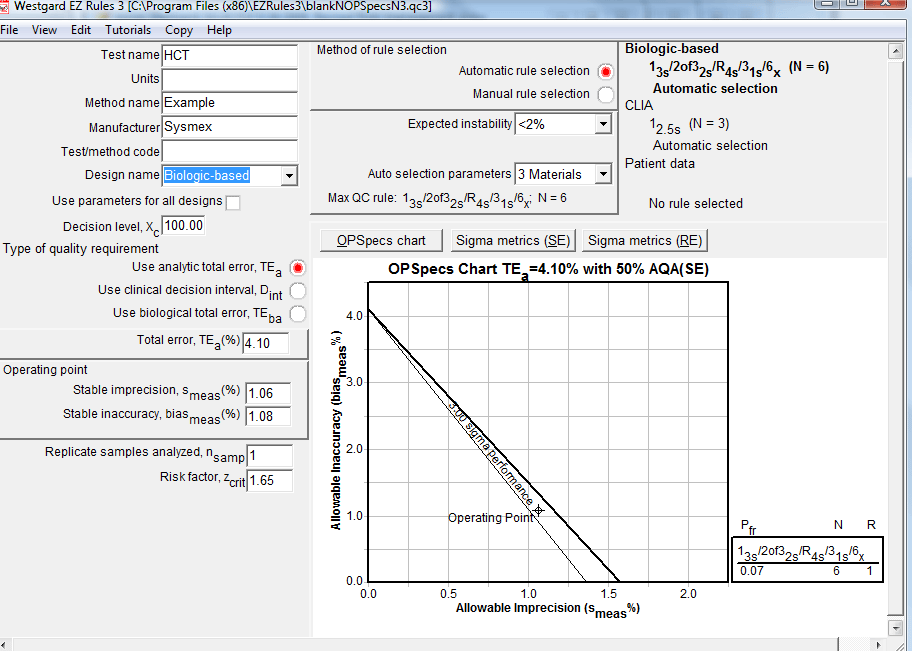
Contrast this with the EZ Rules 3 recommendation using the CLIA quality requirement:
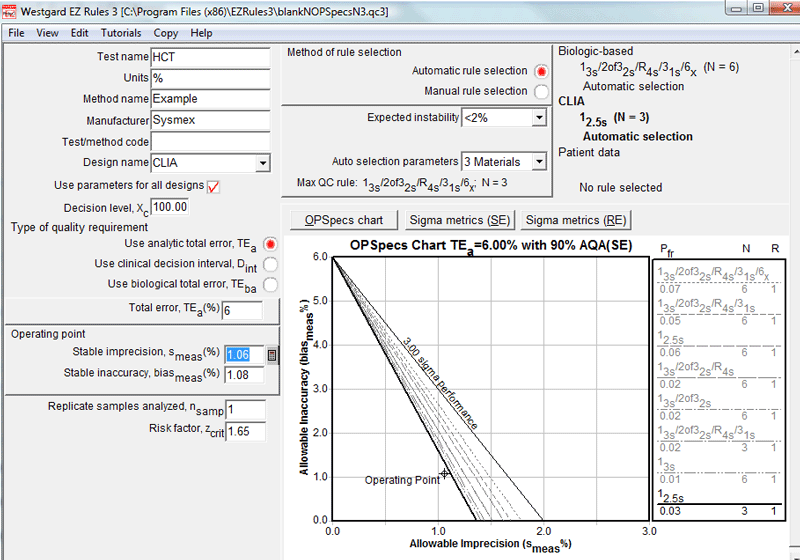
The QC procedure here is more practical: a 12.5s control rule with 3 control measurements.
PLT
PLT is a case where the quality requirements are quite different. The CLIA quality requirement, at 25%, is nearly twice the value of the Biologic-based quality requirement of 13.4%. It's no surprise then that the Sigma metrics are also starkly different. For the CLIA requirement, the Sigma is 6.84. For the Biologic-based requirement, the Sigma is 3.07.
Given the differences, the EZ Rules 3 recommendations should not be a surprise, either:
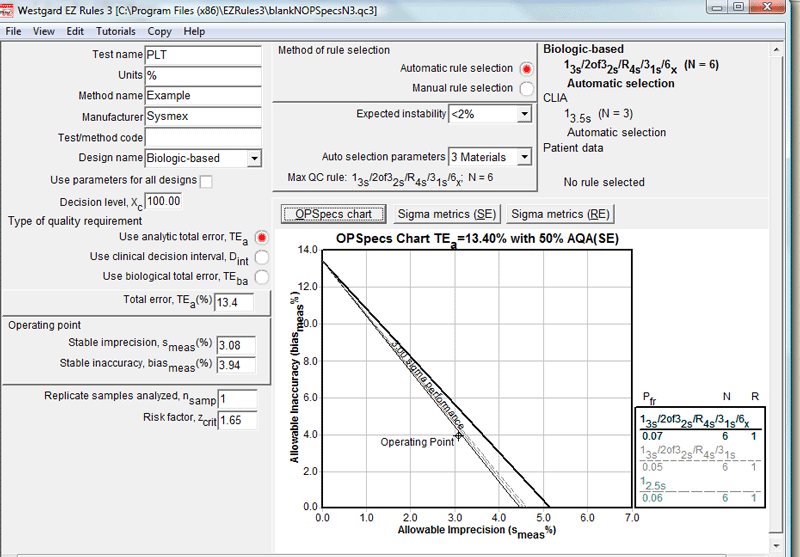
Using the much wider CLIA rule, the QC recommendation is, again, easier to implement:
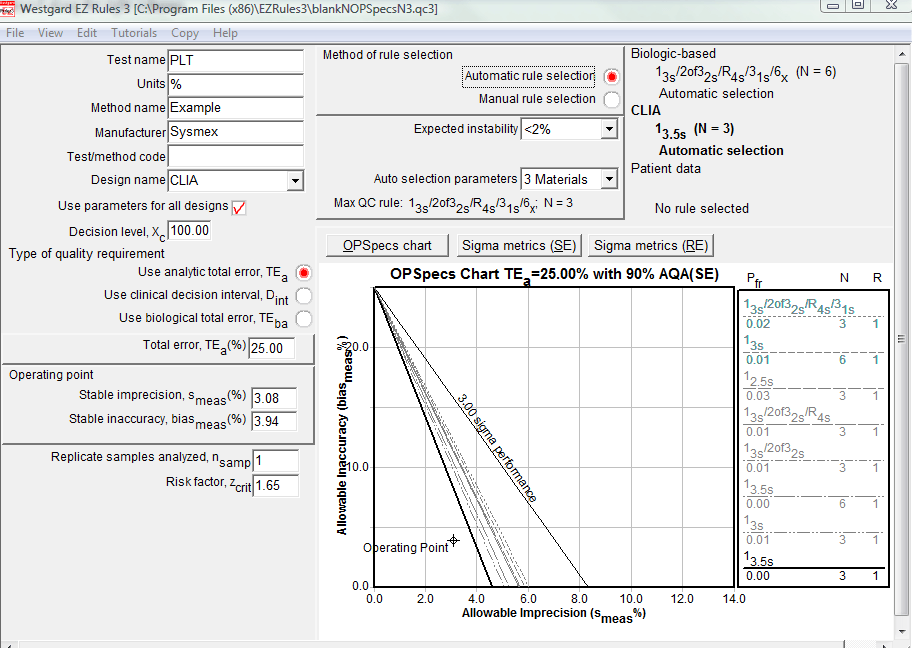
Summary of QC procedure Recommendations
Using EZ Rules 3, we can determine the different QC Recommendations based on the quality requirements. These are displayed below:
| Analyte | Biologic-Based QC procedure recommendation |
CLIA-Based QC procedure recommendation |
| RBC | 13s/2of32s/R4s/31s with N=3 | 13s with N=3 |
| HGB | 13s/2of32s/R4s/31s with N=6 | 13.5s with N=3 |
| HCT | 13s/2of32s/R4s/31s /6x with N=6 | 12.5s with N=3 |
| PLT | 13s/2of32s/R4s/31s /6x with N=6 | 13.5s with N=3 |
| WBC | 13.5s with N=3 | 13.5s with N=3 |
| %Neutrophil | 13.5s with N=3 | --- |
| %Lymphocyte | 13s/2of32s/R4s/31s /6x with N=6 | --- |
| %Monocyte | 13s/2of32s/R4s/31s /6x with N=6 | --- |
| %Eosinophile | 13s/2of32s/R4s/31s with N=3 | --- |
| %Basophile | 13.5s with N=3 | --- |
There is quite a difference between the rules needed for the Biologic-Based requirements versus what is required to achieve CLIA compliance. For a US laboratory, I expect it would be very hard to choose the tighter requirements. Any laboratory making this choice would need to weigh the clinical benefits of controlling those tests to a tighter degree against the costs of quality control.
With the differential parameters, there are no CLIA requirements at all, so the Biologic-Based requirements are all we have to go on. Some parameters will be very easy to control, requiring 3s or 3.5s control limits, while others will require a sort of "Maximum QC."
Yet one more choice to make is how to reconcile the different QC procedures. Given that this is a multitest instrument, at least one decision has to be made that applies to all the tests: how many control measurements will be made with each run? It's probable that the laboratory is going to run just three controls and measure each one once. So all the QC procedures will need to synchronize on that N.
In other words, EZ Rules and QC Design give the laboratory a set of ideal QC procedure recommendations. Now the laboratory must choose a practical set of QC procedures to implement. In some cases, this may mean "under-controlling" a method. For those analytes and parameters, it will be very important to ascertain the extent of the under-control, so the laboratory is aware of how much risk is involved with those methods.
Evaluating the Error Detection with Different QC Procedures
EZ Rules 3 allows you to examine the error detection and false rejection characteristics of any common QC procedure in the context of a specific method, analyte, or parameter. You plug in the method characteristics, use the Manual rule selection, and then select the Sigma metrics chart (previous screens were looking at OPSpecs charts, which do not detail the error detection characteristics of QC procedures).
For example, if you were using a 13.5s control rule with N=3 - a QC procedure that is fine for WBC - but you wanted to know how well that procedure would work for the Lympocyte parameter, you would see the screen below:
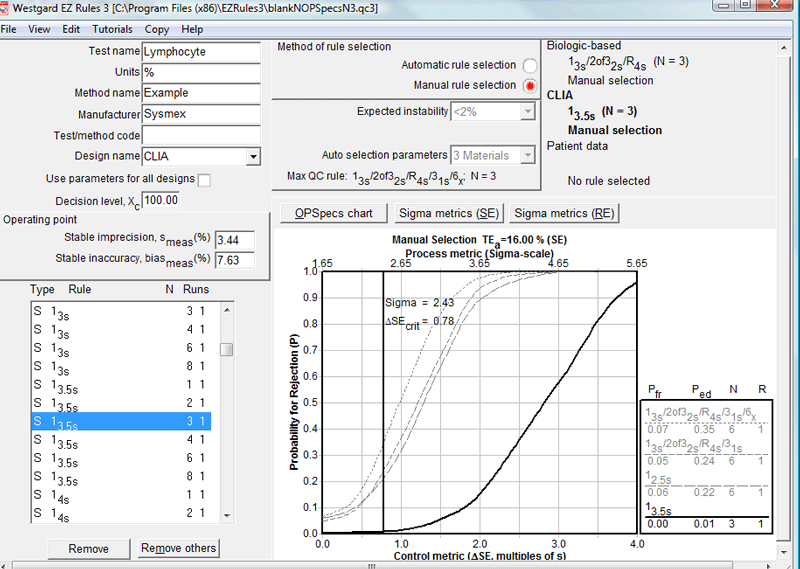
On the right side of the graph, in the key, you see two columns of probability numbers associated with each control rule. The first number is the probability of false rejection. The second number is the probability of error detection. So, in this case, a 13.5s control rule with N=3 will have basically no false rejection, but it will also have basically no ability to detect when a medically important error occurs in the lymphocyte method.
If a more robust QC procedure is used, the numbers improve, as shown below:
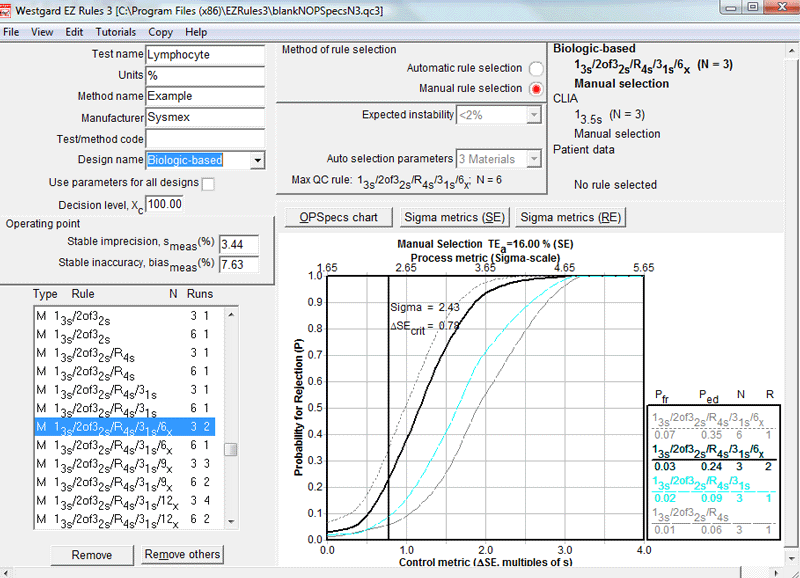
While the approximately 26% error detection is far from ideal, it's a lot bigger than 1%.
Here's where Risk Management techniques make a useful complement to the QC Design process. Using a selected QC procedure, we have attempted to mitigate the risk of a medically important error for a method. But the mitigation does not have the ideal detection power, so there is some "residual risk" left over. How the laboratory - and the clinician - handle and share that residual risk is the next step in the Total Testing Process.
Conclusion
This study shows the scope of decisions that a laboratory must make when it decides to perform QC Design. First, the laboratory must choose which quality requirements to use. It's clear that the choice of the goal has a huge impact on the next step in the QC Design process: choosing QC procedures. When a laboratory chooses QC procedures, it often has a great deal of context to reconcile with the ideal choice. With multitest instruments - the reality for most testing - the laboratory must pick a number of controls that represents the average need of the tests. So if one particularly bad performing parameter requires more controls, but all the other parameters do not, it's realistic to assume that the laboratory will run fewer controls than needed for that method. What's important, in these cases, is to acknowledge the decisions and the tradeoffs, and determine the extent of the risk.
