ISO
ISO-1: Basics on ISO Standards
What is ISO? What is ISO accreditation? Guest authors Daniel Perigo, R.Ph. and Rogerio Rabelo, MD, FACB, present the basic ideas and concepts of ISO standards and how they apply to the clinical laboratory.
- 1. Basic Concepts
- 1.1 Accreditation and Certification
- 1.2 Quality Control vs. Quality Assurance
- 2. ISO Standards
- 3. What you can expect (and not expect) from ISO standards
- 4. A final clarification on our perspective and approach
- References
- About the authors and their institution
See the introduction by Dr. James O. Westgard, Ph.D., FACB
Accreditation processes are widely known in clinical laboratories and medical services worldwide. CAP, JACHO, COLA and other institutions have a very well established know-how in the field. But the ISO System is catching the attention of more and more labs in the last few years. One of the most recent ISO publications – ISO15189:2003 – was developed as a guideline for the implementation of quality systems in medical (clinical) laboratories, complementing the ISO9001:2000 international standard. But what are the differences between these processes? Is laboratory accreditation the same as ISO certification? Are ISO standards really valuable for medical laboratories?
This essay has the intention to present some basic concepts related to ISO System – making a clear distinction with accreditation processes - and to clarify the approach for applying ISO standards in clinical laboratories.
1. Basic concepts
Some concepts must be clearly understood to gain the proper perspective on the ISO approach:
1.1 Accreditation and Certification
The word accredit is related to the same root word as credible, or believable. An accredited institution is one that can be believed. Accreditation can be defined as a voluntary process by which organizations are certified by an independent company that they comply with specific requirements. It is a means of showing confidence in an organization’s performance. Usually, accreditation processes are more focused on the technical aspects of the services provided.
In the ISO system, the independent certifying company is the one that is accredited, meaning that this company must be checked and recognized by other specialized authority bodies in order to be considered capable to certify other companies. The process by which a company is audited by the certifying body is called Certification and compliance is usually demonstrated by written assurances (the certificates). So, for ISO purposes, companies that were found to meet the requirements specified in international standards are called certified, and not accredited, since the certifying company is the one that is supposed to be accredited.
1.2 Quality Control vs Quality Assurance
Quality control (QC), according to the definition from some accrediting bodies, is a process where known samples are tested routinely, in a systematic way, in order to confirm the reliability and precision of analytical procedures. QC can be considered part of the operational control of processes, being extremely useful for detecting and correcting real and potential deviations.
Quality assurance (QA) is a process to aid companies in improving the reliability, quality, and the overall performance of the organizations, helping them to develop a systemic approach of processes and interfaces. Specific standards usually divide QA in three phases: pre-analytical (pre-examination procedures), analytical (examination procedures) and post-analytical (post examination procedures).
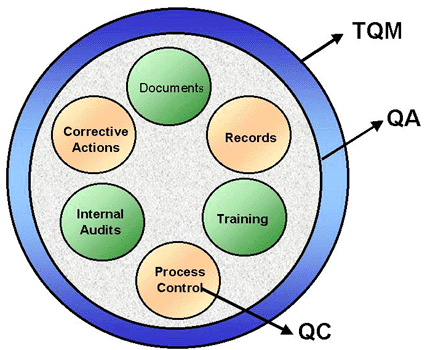
Understanding Quality Assurance as a major and wide system allows us to see QC – and its more specific focus - as part of QA, although the opposite is not true. QA, in turn, can be seen as part of a wider system called Total Quality Management (TQM). TQM can be defined as the use of quality concepts in every process of a company. It states that the different areas have responsibilities in assuring and maintaining quality as a “work philosophy”, a “life style” for business purposes.
In fact, QC, QA and TQM can be considered evolutionary steps of the Quality concept. The ISO 9001 standard, as a Quality Management System, also went through these steps: The old 1994 version (ISO 9001:1994) used a QA-related approach, while its most recent version, the current ISO 9001:2000, has adopted a TQM-related approach.
2. ISO Standards
Although very well known in manufacturing industries, ISO standards are relatively new in healthcare and service organizations. This is one of the reasons why ISO guidelines had been labeled as “product manufacturing standards” for a long time. As the certification of different organizations became more common, its applicability in services and, more specifically, in clinical laboratories, was finally understood.
The International Organization for Standardization, or ISO, is a non-governmental organization with members from international institutes of more than a hundred countries, coordinated by a central office in Geneva, Switzerland. The main purpose of ISO is to develop standards internationally recognized.
The word ISO originated from the Greek isos, meaning equal. That expresses the desire of the institution in developing standards intended to be useful for industries and business organizations of all sizes and types. According to ISO, its idea is “to contribute to making the development, manufacturing and supply of products and services more efficient, safer and cleaner” (ISO Web site: www.iso.org).
ISO standards are developed by specific committees and contain a series of requirements and specifications usually presented in numeric itens. When a committee decides to set up a new standard, some steps must be followed until the final publication:
- DIS (Draft International Standard): after discussion, an initial draft of the standard is circulated among ISO members for consensus
- FDIS (Final Draft International Standard): receiving a favorable feedback, comments are consolidated and the document circulates again as a final draft.
- Standard: after approval, the final text is published.
These standards can be acquired directly from ISO or its member institutes and other authorized organizations.
The standards can be used as guidelines for orientation or adopted as rules to be followed. They are classified as generic or specific.
Generic standards – used for certification purposes: there are two main generic standards: ISO9001: 2000 and ISO14001: 1996, which contain essential criteria for implementing quality and environmental management systems, respectively. As previously described, these standards are intended for certification of companies of all sizes and types, which explains why they have a generic approach. Another characteristic is that both are management standards and must be used by top management as a tool to achieve the expected results of the organization by improving customer satisfaction and reducing negative environmental impacts.
A very important point is that these ISO certification standards are not product standards. They are more concerned about management and processes and do not contain requirements for specific products. Even supposing that a controlled process can lead to a good product - which is not always true - they cannot be considered product guarantees.
Specific standards – usually guidelines for orientation: they are not intended for certification purposes and have a more technical and specific approach. This is the reason why they can be used as guidelines for organizing and standardizing processes in order to achieve better results. They are very useful for understanding how the requirements of a generic certification standard can be applied to a specific activity. ISO15189: 2003 - Medical laboratories: particular requirements for quality and competence - is one example of specific standard that can even be adopted by international bodies for accreditation purposes.
3. What you can expect (or not expect) from ISO standards
Do not expect specific requirements from certification standards. Use them as general guidelines about “What to do”. NCCLS and other specific and technical standards will help you with the “How to do” part of the process.
Do not expect good results without top management involvement. Remember they are management standards intended to improve the overall performance of an organization.
Do not expect good results without people involvement. A system where people are not an integral part will be seen mainly as “more bureaucracy and papers to fill out....” and will not be considered a priority in the daily routine.
Do not expect good results if an ISO system is implemented only for marketing purposes – the money spent will not be worthwhile unless there is a serious intent to improve the quality of the services provided.
Expect good results when an ISO system is implemented as a tool for continual improvement and process standardization.
Expect good results if the ISO system reflects and is aligned with the organization’s philosophy and mission.
4. A final clarification about our perspective and approach
There is no “magic formula” for ISO certification. Each organization must analyze the requirements and find the best way to achieve compliance. Always remember that all the knowledge that is built into your laboratory should be used to meet the ISO requirements.
Keep in mind that organizations should be certified by generic standards. This is exactly the way the ISO system can be used as the basis of more specific accreditation standards and other management tools. Just imagine the ISO system of standards as the big umbrella shown below, able to cover more than one person, approach, strategy, or set of quality management tools and guidelines. That perspective is the key to taking the best of everything to make your laboratory the best.
And remember that the fact of being generic doesn’t invalidate the idea that ISO can provide organizations with a reliable quality management system that assures continual improvement.
To be continued…
References
ISO 9000: 2000, Quality management systems - Fundamentals and vocabulary (this standard can be acquired from ISO website – www.iso.org).
ISO 9001: 2000, Quality management systems – Requirements (this standard can be acquired from ISO website – www.iso.org).
ISO 9004: 2000, Quality management systems - Guidelines for performance improvements (this standard can be acquired from ISO website – www.iso.org).
ISO 14001: 1996, Environmental management systems - Specification with guidance for use (this standard can be acquired from ISO website – www.iso.org).
ISO 14004: 1996, Environmental management systems - General guidelines on principles, systems and supporting techniques (this standard can be acquired from ISO website – www.iso.org).
ISO 15189: 2003, Medical laboratories: Particular requirements for quality and competence (this standard can be acquired from ISO website – www.iso.org)
ISO Basics: http://www.iso.org/iso/en/iso9000-14000/basics/general/basics_1.html [link no longer works]
CAP Laboratory Accreditation: http://www.cap.org/apps/cap.portal?_nfpb=true&_pageLabel=lab_inspections_page
About the Authors and their Institution
Rogério Rabelo, M.D., Ph.D.
Dr. Rabelo is the Management Representative for Fleury Diagnostics’ Integrated Management System (ISO 9001:2000 and ISO 14001:1996). He is also a Medical Consultant in Clinical Chemistry/Medical Biochemistry and a Product Manager (High-volume, Automated Testing and POCT) at Fleury Diagnostics.
He got his M.D. from the Federal University of Goiás, in Brazil and his Ph.D. from the University of São Paulo, Brazil, with a joint research project developed at the Lady Davis Institute for Medical Research, McGill University, Canada. His post-doctoral training in Clinical Chemistry/Medical Biochemistry was done in the Department of Medical Biochemistry/Laboratory Medicine, Jewish General Hospital, McGill University, Canada. He is currently doing his MBA at University of São Paulo, Brazil.
Daniel Marques Périgo, R.Ph.
Mr. Périgo is the Integrated Management System (IMS) Coordinator responsible for Fleury Diagnostics’ Quality and Environmental Department.
He is a Pharmacist, graduated by University of São Paulo, Brazil, with specialization in Clinical Assays. He is also certified as specialist in both Quality Management and Productivity (by Vanzolini Foundation, from University of São Paulo) and Environmental Management (by the College of Public Health, University of São Paulo, Brazil).
NOTE: Fleury Diagnostics’ Quality and Environmental Department has only 3 FTEs: Mr. Périgo as the IMS Coordinator and two IMS Assistants, Mr. Daniel P. Santos and Mrs. Adriana S. E. Gonçalves. This small Department is responsible for the operational management of the entire ISO 9001: 2000 and 14001: 1996 System. This is only possible due to the high degree of decentralization of Fleury's IMS, where all departments and every employee are responsible for the maintenance and continual improvement of the System.
Fleury Diagnostics Background: Knowing the Lab to Understand its ISO Quality and Environmental Systems
1. Laboratory and Diagnostic Medicine at Fleury Diagnostics: Strategic Focus
Founded in 1926, Fleury Diagnostics is a comprehensive Diagnostic Medicine Center that offers the most complete menu of tests in Brazil. Since its very beginning, it has been providing humanized and personalized services, always striving to improve the quality of its tests. Over the years, the laboratory has grown and expanded significantly, inaugurating several new facilities in São Paulo City, in other cities in the state of São Paulo and in Brasília and Rio de Janeiro. Fleury’s outpatient facilities are specially designed to provide a pleasant, comfortable environment to the patients. They are operated by highly specialized staff, which delivers fast and friendly service, with personalized and humanized care.
Fleury Diagnostics serves more than 2,500 patients a day and offers more than 2,000 different types of diagnostic tests in a wide variety of specialties like Clinical Pathology, Anatomical Pathology, Radiology, Medical Imaging, Cardiology, Fetal Medicine, Endoscopy, Molecular Genetics and in 30 other medical specialties. All tests performed at Fleury follow strict International standards of internal and external quality control. Fleury is also the main Reference Laboratory in the country, analyzing send-out tests from more than 500 laboratories located anywhere in Brazil.
In order to assure the post-analytical quality of the services provided, Fleury's clinical staff has more than 250 highly specialized physicians, 15 researchers and 300 biochemists, biologists and biomedical professionals, graduated and trained in the best universities in Brazil and abroad. These professionals are always available to provide diagnostic consultancy services to physicians and clients. This service can be easily and swiftly accessed with a phone call or through the Internet. A call center for customer service is open 24 hours a day, answering more than 6,000 calls a day, providing information on all the services available, scheduling exams and directing all questions to Fleury’s medical staff.
Since 2001, Fleury’s technical, educational and administrative activities have been centralized at its new headquarter – a 140,000 square-feet complex in São Paulo City. Endowed with the most modern equipment, this complex has given Fleury an even greater technical excellence, better quality of life in the working environment and higher productivity. The facility has the capacity to perform 20 million tests per year. In 2003, Fleury Diagnostics created the Fleury Institute, with the mission of managing and implementing its educational, social responsibility and research & development projects.
To make the day-to-day lives of patients and doctors easier, Fleury provides a modern result retrieval system on the Internet that can also be used even for imaging tests. This system was built with the best confidentiality and safety protocols available (as safe as Internet Banking). Fleury Diagnostics was the first laboratory to make test results available on the Internet (1997), and the registered patients will receive an e-mail alert as soon as their results are ready. One can also get the test result reports at Fleury’s facilities or have them delivered by mail, express delivery, fax or motorcycle courier. Several services are provided to doctors and clients on Fleury’s web site (www.fleury.com.br) like online scheduling, detailed information on all tests available, research papers and multimedia courses.
2. Fleury Diagnostics mission and Integrated Management Policy: Patient Focus
Fleury Diagnostics’ mission is to develop and provide medical services and diagnostic procedures with ethics and technical excellence, to fulfill the needs of the clients.
Based on this mission, Fleury Diagnostics commitment to quality is fully focused on the patient needs and it is always striving for the best possible service and the excellence of test results. As a diagnostic company so concerned with the quality of the services provided, Fleury needed a formal system to demonstrate its capability to meet customer satisfaction for the services offered. It has chosen to implement the ISO Quality Management System because this was exactly the ISO 9001 goal. In 1999, Fleury Diagnostics became the first laboratory in Latin America to be granted the ISO 9001:1994 certification, with a very successful implementation due to the effort and involvement of all employees. In 2002, it reinforced this commitment to quality and also demonstrated its commitment to the environment. This was achieved by the implementation of an Integrated Management System (IMS) based on the ISO 9001:2000 Quality Management System and the ISO 14001 Environmental Management System, certified in August of 2002.
As a result, the Integrated Management Policy expresses Fleury’s commitment to the quality of services and respect to the environment:
“To perform and develop, with technical excellence and ethics, customer services and medical procedures, committing itself to the continual improvement of the services provided by:
Implementation and maintenance of the Integrated Management System
Respect to the environment and prevention of pollution by complying with the applicable legislation and by treating the waste produced.”
This approach is guided by five principles:
- Provide efficient management tools that contribute to continual improvement
- Develop customer services in an efficient and respectful way, in order to answer the needs of the clients and exceed their expectations
- Perform, with technical excellence and ethics, diagnostic and therapeutic procedures
- Reduce the environmental impact of the medical procedures and services by the rational usage of natural resources and complying with legal requirements
- Ensure continuous improvement of the Integrated Management System by setting up goals and effectively monitoring them.