Sigma Metric Analysis
Erba XL 640 in Kolkata, India
A recent study looked at the analytical performance of a Erba XL 640 in Kolkata, India. A trend of poor performance is starting to emerge with the Erba analyzers. Is it the instrument? Or is it where the instruments are operated?
An Erba XL 640 in Kolkata, India
April 2026
Sten Westgard, MS
[Sigma quality score: +1 for bias comparison through EQA program; +0.5 bias measured over long term period; + 0.5 control levels measured near critical decision levels; +0.5 imprecision measured under intermediate precision conditions. Final score: 2.5 = Optimistic.]
A recent study from the Clinical Biochemistry Service Laboratory of the College of Medicine and Sagore Dutta Hospital in Kolkata, India, examined the performance of the Erba XL 640 clinical chemistry analyzer, specifically just 6 routine enzymes:
Application of six sigma metrics of routine enzymes for assessing the quality performance of biochemical analytes in the medical laboratory - A cross-sectional study. Sidhiraj Ghosh, Angshuman De, Satwika Sinha. Asian Journal of Medical sciences. January 2026: Voly 17, Issue 1, pp 27 - 33.
Calculating Sigma metrics from routine laboratory performance data.
The imprecision and bias of the analyzers
"Data from internal QC and EQAS reports for six enzymes run on a biochemistry autoanalyzer, fro mApril 2023 to September 2023, are included in this study....[T]wo IQC levels of normal and high value are run in the clniical chemistry autoanalyzer daily....The lyophilized external QC sample sent from CMC Vellore is reconstituted before the 10th day of every month and run in the clinical chemistry autoanalyzer. the EQAS result is uploaded online before the 20th of every month. The EQAS results are received at the beginning of the next month....The bias percentage for each test was calculated from the laboratory and peer group mean result retrieved from the EQAS reports."
Performance goals used in this study included "TEa guidelines from Clinical Laboratory Improvement Amendments, the Royal College of Pathologists of Australasia, and the European Federation of Clinical Chemistry and Laboratory Medicine."
For our analysis, we are focusing on just the CLIA 2025 goals, with the exception of the analyte lipase, which is not directly regulated by CLIA. For this analyte, we use the biological variation database minimum analytical specification.
| Analyte | % Bias | % CV | Sigma |
| Alk Phos | 4.8 | 3.7 | 4.11 |
| Alk Phos | 4.8 | 4.5 | 3.38 |
| ALT | 7.3 | 7.5 | 1.03 |
| ALT | 7.3 | 6.2 | 1.24 |
| Amylase | 13.4 | 6.2 | 1.06 |
| Amylase | 13.4 | 5.8 | 1.14 |
| AST | 6.3 | 7.2 | 1.21 |
| AST | 6.3 | 5.1 | 1.71 |
| GGT | 4 | 8.1 | 1.36 |
| GGT | 4 | 6 | 1.83 |
| Lipase | 7.5 | 8 | 1.41 |
| Lipase | 7.5 | 4.7 | 2.40 |
One immediately notices that 5 of the 6 analytes are below 3 Sigma, traditionally considered the minimum acceptable performance.
Graphic Display of performance by Normalized Method Decision Chart (NMEDx)
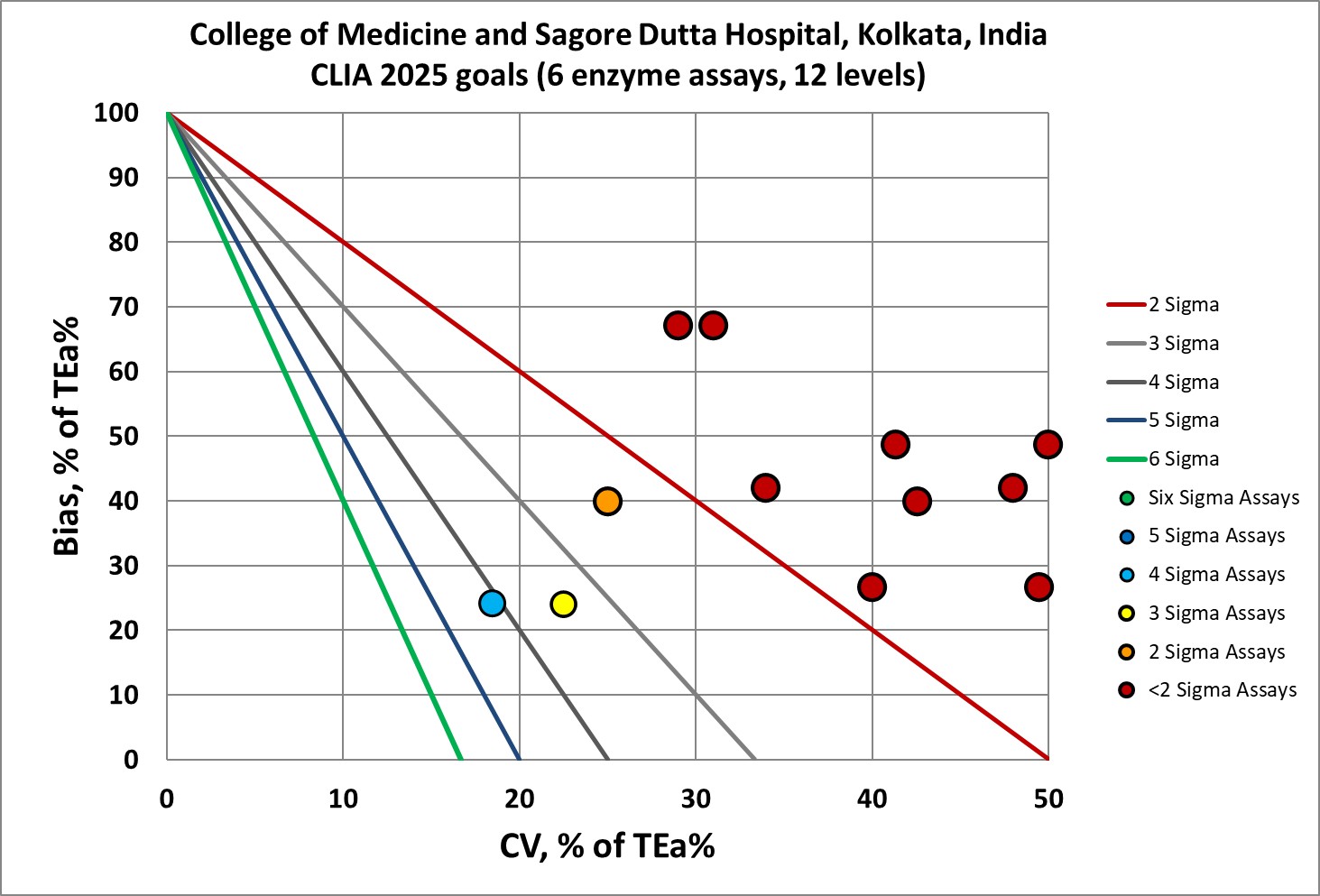
Not a pretty picture.
Most of this performance is bad. It would be considered unacceptable in the US. Is a lower standard of quality acceptable for India?
Conclusion
The authors of the study concluded: "Therefore, parameters that have a sigma value <3 must be reported carefully to identify the root cause and take corrective action to improve the quality of reports in the laboratory. Furthermore, revision in the daily work load division is also recommended by Westgard sigma rules. For analytes with sigma values <3, a change in the frequency of daily runs was also required in our laboratory. Therefore, by using the Standard QC selection tool and Westgard sigma rule, we can select the right QC (frequency of internal QC and number of controls) for the instrument and laboratory."
This is the third study we've seen with poor performance from Erba. The previous two studies are listed here:
Erba XL 640 in India - Westgard QC
Erba EM 360 autoanalyser Sigma-metric analysis - Westgard QC
If we use CLIA 2025 standards, the judgment is far harsher. It begs the question: is there a problem with the lab, the instrument, or both? The rosiest scenario would be a problem with QC handling by the individual laboratory, something that could be corrected with training. On the other hand, the worst scenario would be that the instrument has been engineered such that high imprecision is the expected outcome. That is, the instrument is not capable of acceptable performance. Further research is needed to determine if this is true.