Sigma Metric Analysis
Sight OLO in UK, Multimode Analysis
Continuing in our analysis of hematology systems, we look at the impact of new CLIA and EFLM goals on the assessment of a Sight OLO in the United Kingdom.
Sight OLO in United Kingdom, multimode analysis
July 2023
Sten Westgard, MS
See the other analyses in this series:
- Beckman Coulter DxC 700
- Abbott Alinity
- Siemens Atellica
- Siemens Atellica in Romania
- Siemens Atellica in Spain
- Siemens ADVIA 2120i
- Roche c501 in Turkey
- Roche c501 in Saudi Arabia
- MicroLab RX-50 in India
- Roche cobas 6000 immunoassays in Turkey
- Sysmex XN 350 in India
- Dymind DH 76 in Bulgaria
- Mindray 7500 in China
- Mindray BS 2000M in China
This Sight OLO data comes from St. Thomas's Hospital in London, United Kingdom:
"Reproducibility was assessed using the three-level OLO Control Kit. Each level of QC was tested on OLO in single rns, three times a day, for 20 days. Test runs were executed by a single operator."
"One hundred and thirteen venous whole blood samples collected in K2 ethylenediaminetetraacetic acid (EDTA) tubes were analysed with both OLO and Unicel DxH 800 analysers to assess the agreement of results between the two methods....To assess method comparability Passing-Bablock regression was applied to each measurand. Slope and intercept coordinates were assessed to verify result comparability."
The use of manufacturer controls are a weakness of this study, as they are less likely to provide an honest, independent assessment of the performance of the instrument. But on the hematology side, it's far less common to use a 3rd party control.
There are three levels that will be analyzed here. So for 13 parameters there will be 39 data points to plot on the Normalized Method Decision Chart. Note that while the EFLM biological variation database contains performance specifications for MCV, MCHC, and all the differential paraments, the CLIA 1992 and CLIA 2024 goals do not. Thus, the graphs for CLIA will only cover 5 parameters and 15 data points.
The TEa goals applied can be found on our Consolidated Hematology Performance Specifications page.
| Parameter | Level | Slope | Y-intercept | Bias% | CV% |
| Hemoglobin | 75.18 | 1.05 | -9.03 | 7.011 | 1.60 |
| 134.53 | 1.05 | -9.03 | 1.712 | 1.50 | |
| 173.03 | 1.05 | -9.03 | 0.219 | 1.40 | |
| Hematocrit | 22.73 | 1.08 | -3.71 | 8.322 | 1.90 |
| 41.13 | 1.08 | -3.71 | 1.020 | 1.60 | |
| 54.75 | 1.08 | -3.71 | 1.224 | 1.60 | |
| MCV | 80.2 | 1.02 | -2.16 | 0.693 | 1.20 |
| 85.9 | 1.02 | -2.16 | 0.515 | 0.80 | |
| 92.76 | 1.02 | -2.16 | 0.329 | 0.90 | |
| MCH | 26.53 | 0.96 | 0.96 | 0.381 | 1.10 |
| 28.11 | 0.96 | 0.96 | 0.585 | 0.90 | |
| 29.31 | 0.96 | 0.96 | 0.725 | 1.20 | |
| MCHC | 315.9 | 0.77 | 73.85 | 0.378 | 1.00 |
| 327.27 | 0.77 | 73.85 | 0.435 | 0.90 | |
| 330.77 | 0.77 | 73.85 | 0.673 | 1.40 | |
| Platelets -Impedance | 85.68 | 0.95 | 14.1 | 11.457 | 6.60 |
| 188.83 | 0.95 | 14.1 | 2.467 | 5.50 | |
| 467.3 | 0.95 | 14.1 | 1.983 | 7.30 | |
| RBC | 2.83 | 1.06 | -0.308 | 4.883 | 2.00 |
| 4.79 | 1.06 | -0.308 | 0.430 | 1.60 | |
| 5.9 | 1.06 | -0.308 | 0.780 | 0.70 | |
| WBC | 2.95 | 1.01 | -0.052 | 0.763 | 4.30 |
| 8.73 | 1.01 | -0.052 | 0.404 | 3.70 | |
| 23.65 | 1.01 | -0.052 | 0.780 | 3.10 | |
| Lymphocytes | 0.69 | 1.05 | -0.016 | 2.681 | 10.50 |
| 2.25 | 1.05 | -0.016 | 4.289 | 5.80 | |
| 6.52 | 1.05 | -0.016 | 4.755 | 5.10 | |
| Monocytes | 0.22 | 0.99 | -0.006 | 3.727 | 18.70 |
| 0.64 | 0.99 | -0.006 | 1.937 | 12.60 | |
| 1.35 | 0.99 | -0.006 | 1.444 | 10.00 | |
| Eosinophils | 0.31 | 1.1 | 0.017 | 15.484 | 16.80 |
| 0.57 | 1.1 | 0.017 | 12.982 | 14.70 | |
| 1.16 | 1.1 | 0.017 | 11.466 | 9.30 | |
| Basophils | 0.31 | 0.71 | -0.003 | 29.968 | 15.00 |
| 0.65 | 0.71 | -0.003 | 29.462 | 12.50 | |
| 1.4 | 0.71 | -0.003 | 29.214 | 9.90 | |
| Neutrophils | 1.42 | 0.98 | -0.024 | 3.690 | 6.50 |
| 4.61 | 0.98 | -0.024 | 2.521 | 4.20 | |
| 13.21 | 0.98 | -0.024 | 2.182 | 3.80 |
Sigma-metrics according to EFLM-derived DESIRABLE performance specifications
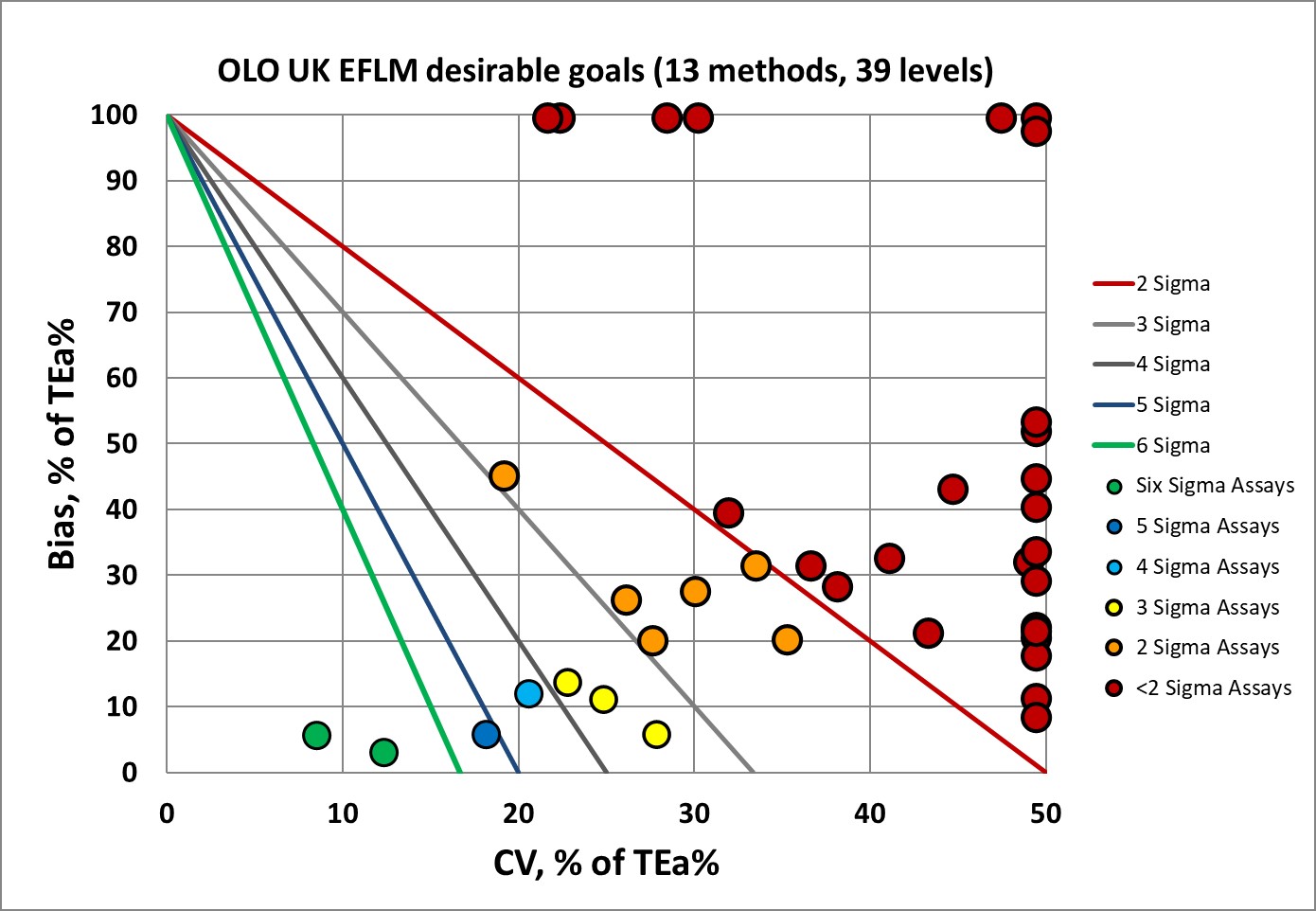
The EFLM desirable specifications used to be the de facto global standard, but have fallen out of favor due to their toughness.
The Sight OLO has over eighty percent of the performance below 3 Sigma. The main driving factor is imprecision that is too high relative to the performance specifications.
Not surprising then, that EFLM recommended lowering the standards.
Sigma-metrics according to EuBIVAS-derived MINIMUM performance specifications
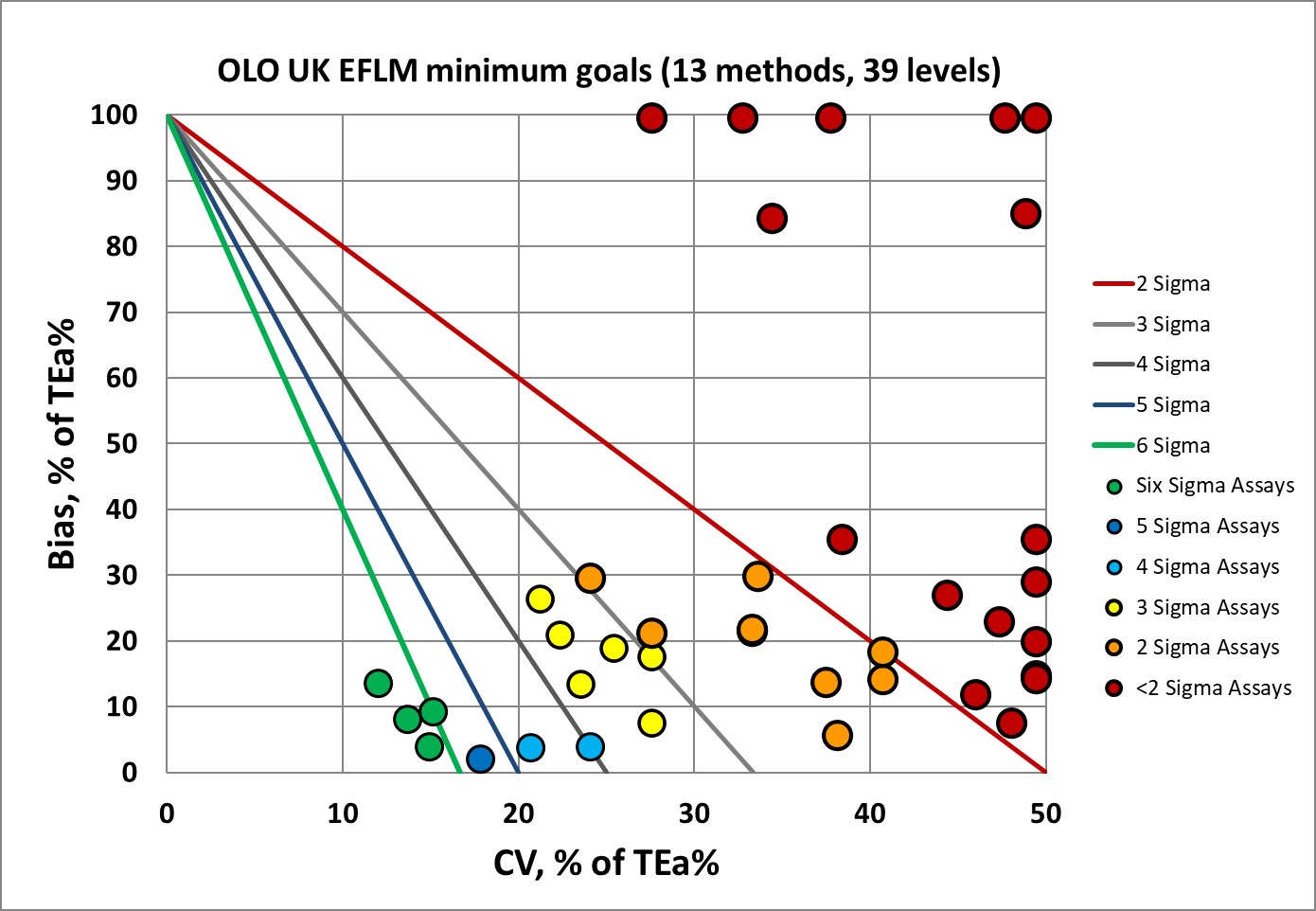
There isn't that much of an improvement, with still two-thirds of performance below 3 Sigma, and less than one-third of that performance below 3 Sigma.
Here's one of the most interesting new aspects of CLIA's new 2024 goals. Are they more or less demanding than EFLM goals?
Sigma-metrics according to CLIA 2024 performance specifications
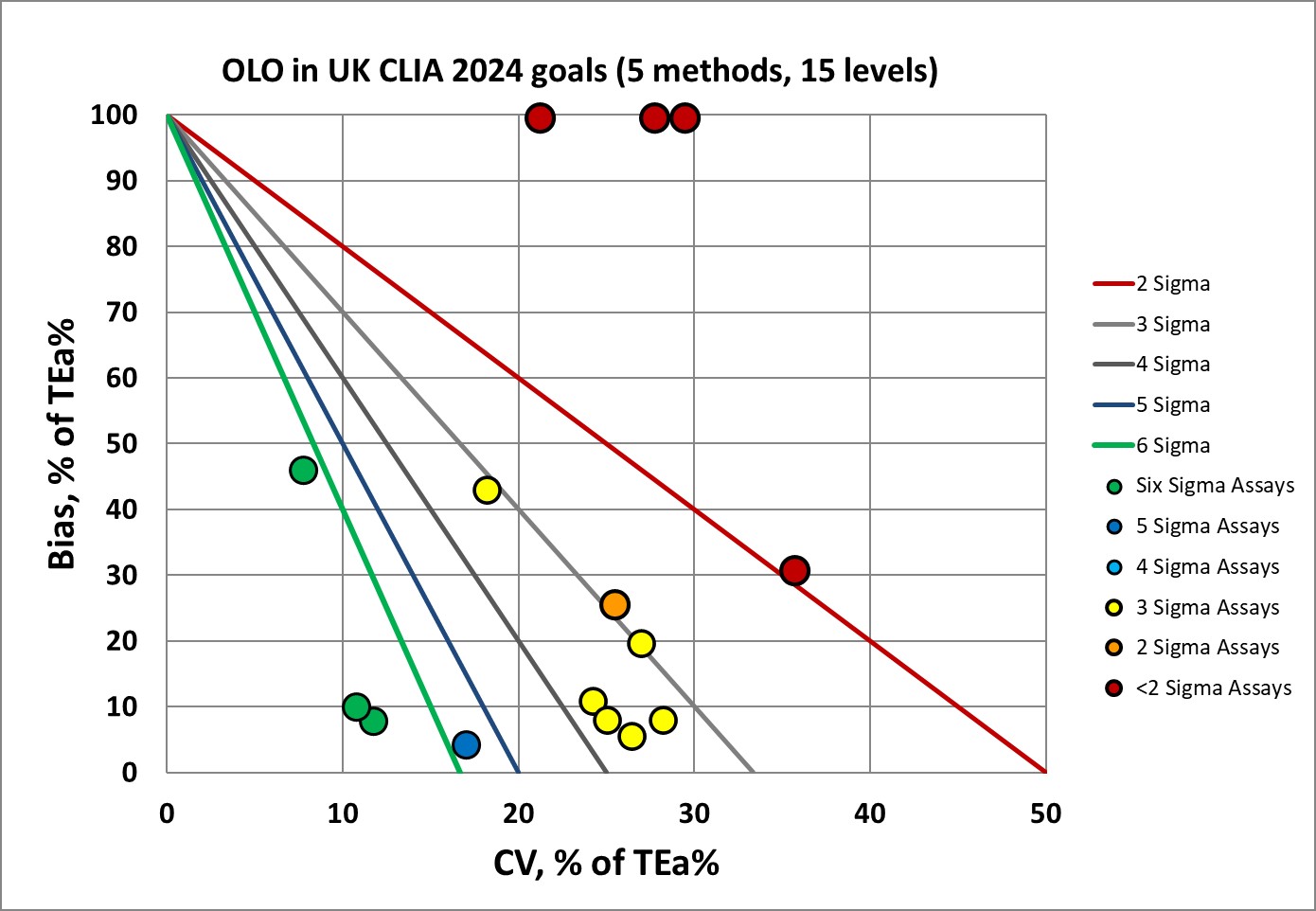
The CLIA 2024 goals are a bit easier on the OLO, with only 40% below 3 Sigma.
Sigma-metrics according to CLIA 1992 performance specifications
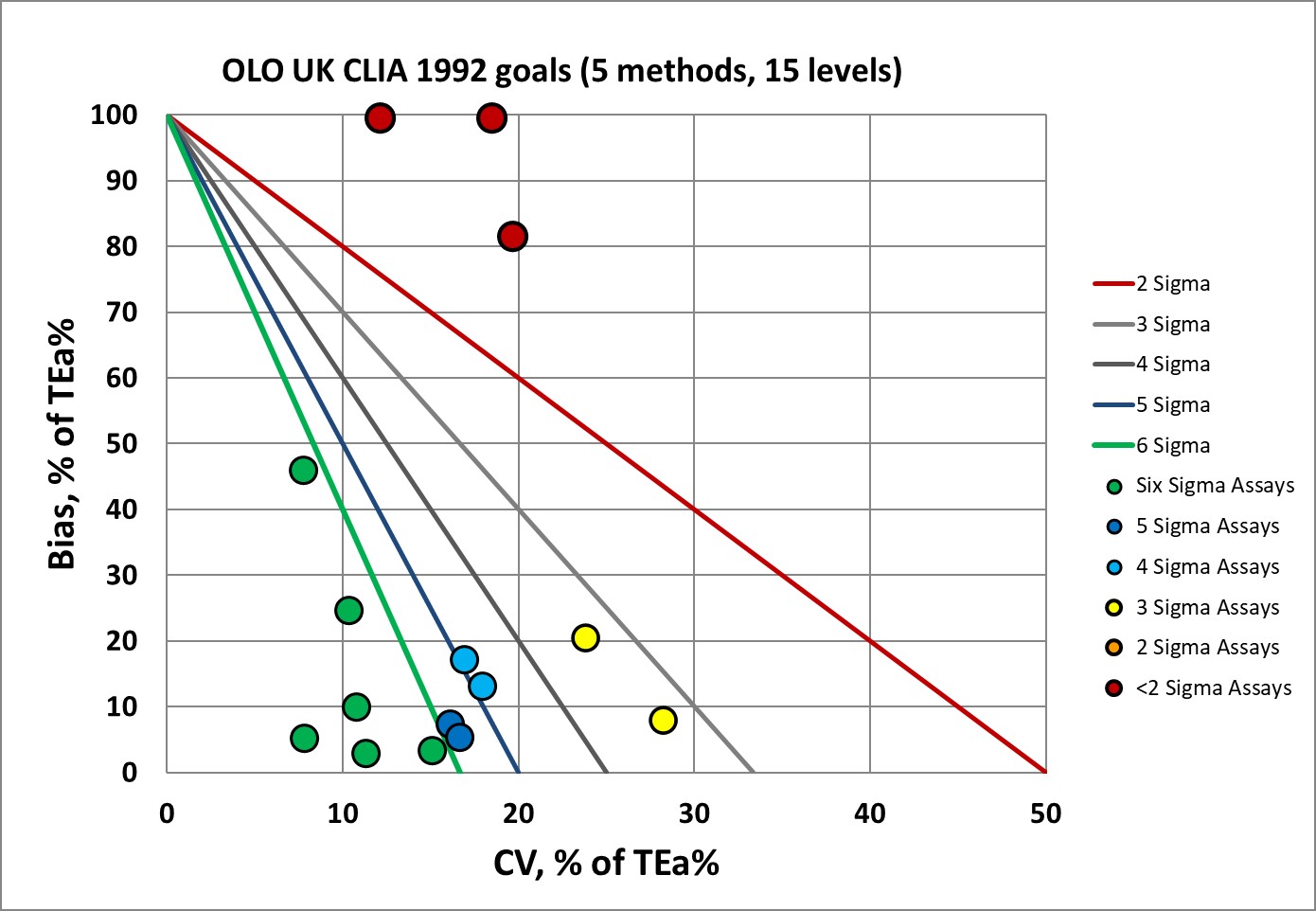
If we apply the CLIA 1992 goals, it's even more favorable. The most number of 6 Sigma analytes occur when these performance specifications are used. But no set of goals gives the OLO a clean bill of health.
Conclusion
Even with possibly the most favorable assessment (from their own controls), the Sight OLO has a lot of difficulty hitting any set of targets. With the most recent goals (CLIA 2024) that have been imposed, it's possible to explain away the failures by noting that the tighter goals are an over-correction, an attempt to make up for decades of stagnation with the CLIA PT goals. It's possible to argue that a comparison of the OLO against the DxH may not be the best choice, if OLO is truly a different methodology, but the main problems dragging down the Sigma metrics are imprecision, not bias. It's also possible to look at the EFLM biological goals (either minimum or desirable) and note that hardly any platform on the market has been able to successfully hit them with consistency. Indeed, there's growing evidence that the current set of goals for hematology, from both CLIA and EFLM, are not in synch with today's analyzers. But if the OLO can't hit the goals from 30 years ago, that's not an encouraging sign for an instrument introduced in the 2020's.
